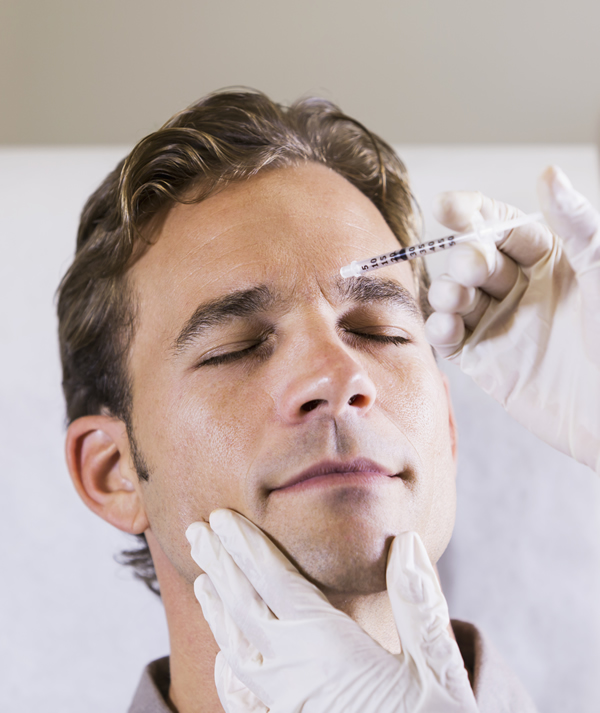
Aesthetic
ReViVox® is a safe, low dose, formulation of pyridostigmine under development as a reversal agent for off-target muscle paralysis after botulinum-Type A toxin injections. In the cosmetic application, ReViVox® will initially be developed as an injectable formulation. As a follow-on development, the drug may be formulated for topical delivery (e.g. ointment).
Development Path for ReViVox®
- Preliminary non-clinical Proof of Concept data available with dose-finding and drug formulation work in progress with CRO partners at study and stability; conduct IND-enabling studies, drug CMC development, and stability, hold pre-IND meeting with FDA. .
- File IND with FDA, 505b2 regulatory pathway
- Human pilot study, 15 subjects (estimated)
- Ph1/2, 125-150 human subjects (estimated)
- Ph 3 Pivotal Study & Submission to FDA
- Estimated time to completion: 2.5 years